Received: 2025-01-04
Accepted: 2025-03-13
Published: 2025-04-01
Pages: 63-72
Antibody-drug conjugates (ADCs) are a novel class of biopharmaceuticals. They combine the specificity of monoclonal antibodies with the potency of small-molecule drugs. This review traces the evolution of ADCs from conception to clinical application and focuses in greater detail on three critical aspects: antibody selection, linker technologies, and payload optimization. We analyse the mechanism of action, shifting emphasis to targeted delivery and controlled release of cytotoxic agents. It discusses FDA-approved ADCs in oncology and promising candidates in clinical trials while exploring emerging applications in autoimmune and infectious diseases. Recent technological breakthroughs in site-specific conjugation and novel payload development are placed alongside ongoing challenges including drug resistance and toxicity management. We evaluate combination therapy approaches and the integration of ADCs into personalized medicine frameworks. This comprehensive review will be a treasure chest for researchers, clinicians, and pharmaceutical professionals to deal with constantly changing areas of antibody-drug conjugates.
Antibody-drug conjugates are a new form of biopharmaceutical in which the specificity of monoclonal antibodies is combined with the potency of cytotoxic drugs, revolutionising targeted therapy, especially in oncology. The ADC links a monoclonal antibody, a cytotoxic payload, and a linker connecting the payload to deliver toxic payloads directly to antigen-expressing cells with reduced damage to normal tissues [1].
The concept, born from the early 20th century "magic bullet" by Paul Ehrlich, found its form in the 1970s with hybridoma technology [2]. Early ADCs of the 1980s had problems with immunogenicity and low potency [3]. Essential changes began at the millennium; gemtuzumab ozogamicin was approved in 2000,followed by brentuximab vedotin in 2011 and ado-trastuzumab emtansine in 2013 [4].
The ADC field has exploded at a fantastic pace. In fact, by 2024, over two dozen FDA-approved ADCs will have been established, and many more are under clinical development [5]. The field continues to be steered by innovations in antibody engineering, linker design, and payload selection [6]. This review uncovers the composition and modes of action and highlights existing uses in cancers and other diseases, recent technical progress, and challenges facing this dynamic field of targeted therapeutics. In this respect, the following discussion will aim to paint an overall picture of current situations and prospects in ADC technology and how it can be poised to change treatment programs across numerous diseases.
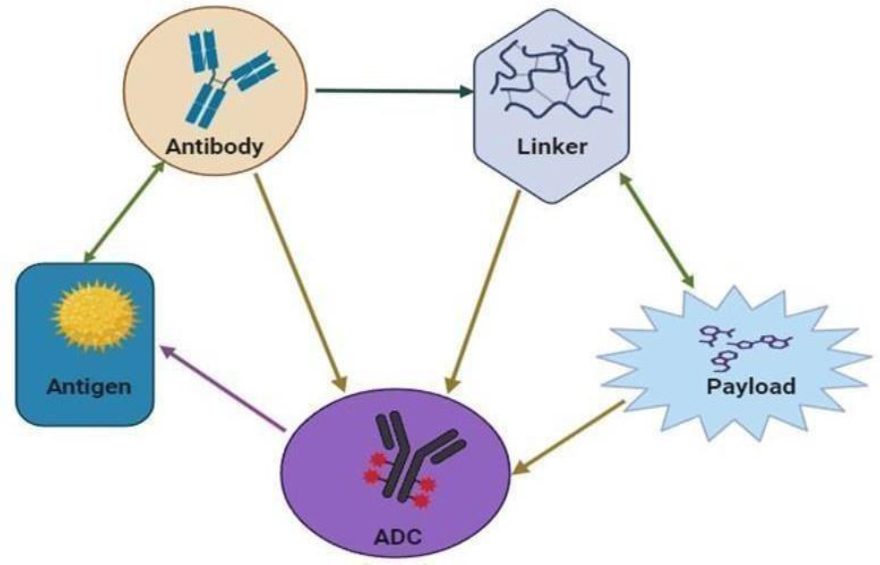
The diversity of selection processes, advanced engineering of antibodies, innovative linker technologies, and judicious selection of payloads embody the core constituents that form the structural framework and necessary components of antibody-drug conjugates, as shown in Figures 1 and 2. The process of selection and engineering of antibodies is very complex. It involves the identification and selection of monoclonal antibodies that are further humanized for compatibility with the human immune system, as well as affinity maturation with meticulous attention to enhance the binding strength of antibodies. Additionally, the Fc region is altered to improve effector functions, such as ADCC and CDC [7].
Linker technologies comprise a wide range of cleavabl as well as non-cleavable linker types; the former cleavable types include hydrazone, peptide, and disulfide linkers that allow the timed- release of the cytotoxic payloads within the target cells, while the later non-cleavable types, such as thioether linkers offer stability in circulation, together with other site-specific conjugation strategies to improve the target specificity and therapeutic index [8].
The most important decision regarding payloads is indeed the judicious choice of heavily potent cytotoxic agents, such as auristatins and maytansinoids, which have demonstrated potent antitumor activity; however, it also involves DNA-damaging agents acting to impede cancer cell proliferation and novel payloads deployed through new mechanisms of action to compensate for and surpass resistance toward better and enhanced therapeutic results [9].

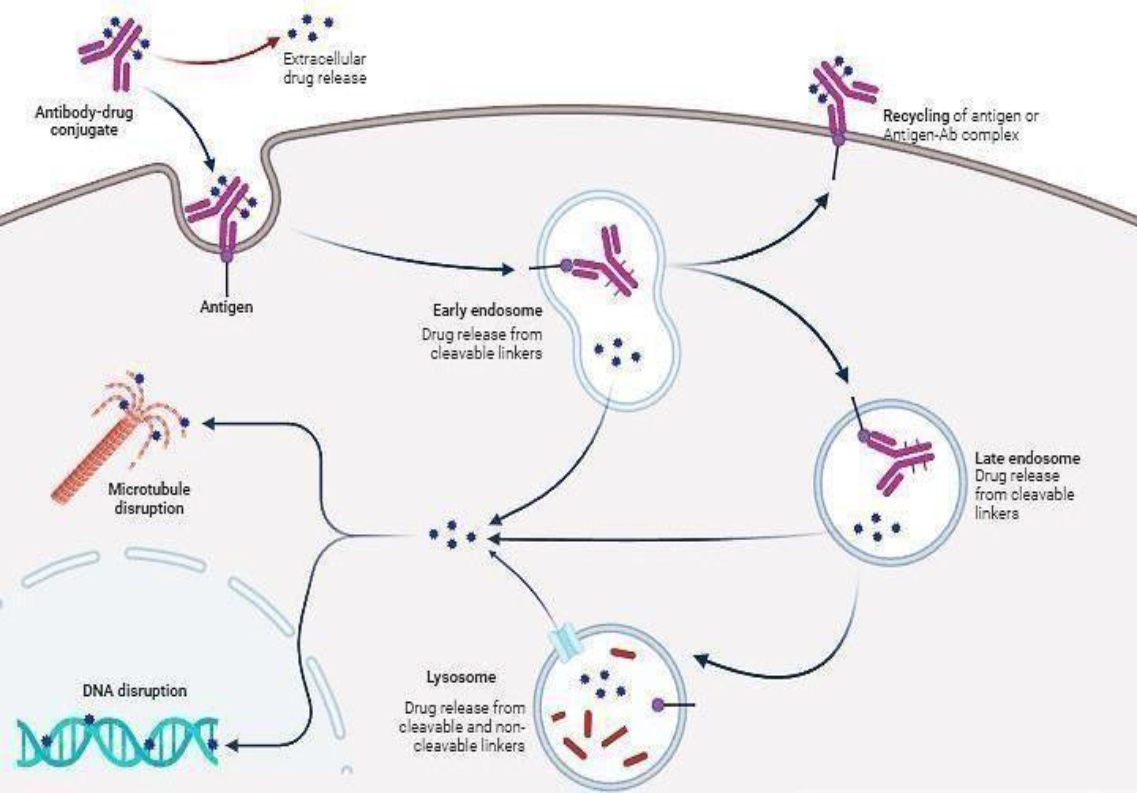
ADCs use a precision-guided mechanism through which the antibody first binds to specific targets on cancer cells, resulting in cellular internalisation that releases potent drug payloads inside cancer cells. This process of targeted drug delivery allows cancer cells to be selectively killed while sparing much of the surrounding healthy tissue, thereby making ADCs a significant therapeutic approach in modern cancer treatment, as shown in Figure 3.
An ADC is targeted to its site of action through its antibody component, engineered to recognise and bind specifically to antigens predominantly expressed on the surface of cancer cells while being minimally present in healthy tissues. Well- characterized targets have emerged in clinical applications: HER2 is commonly associated with breast cancer, CD30 is found in lymphomas, and CD33 in leukaemias. This selective targeting capability creates the essential therapeutic window that makes ADCs effective as cancer treatments [10].
Following target recognition, the ADC undergoes a sophisticated cellular entry process. Receptor- mediated internalisation is initiated upon binding to its specific antigen on the cell surface. This process involves invading the cell membrane around the ADC-antigen complex, forming an endosome. Then, the newly formed endosome migrates through the cytoplasm and finally fuses to lysosomes. Even though internalisation efficiency is highly dependent on the targeting antigen and the particular characteristics of the antibody, optimal targeting is essential for correct internalisation and therapeutic effect [11].
The mechanism of payload release works through two different strategies involving linkers: The design of cleavable linkers is to maintain stability in the bloodstream but to specifically degrade in intracellular conditions, either due to lysosomal acidic pH, certain cellular enzymatic activities or the reducing environment of the cytoplasm. Non-cleavable linkers require comprehensive degradation of the antibody within the lysosomal compartment, leading to the release of the payload still attached to the residual amino acid. The choice between these linker types significantly impacts the ADC pharmacokinetics, efficacy, and toxicity profile [12].
The payload cell-killing mechanism primarily follows two significant strategies. Microtubule inhibitors, such as auristatins (MMAE) and maytansinoids (DM1), act through mitotic blockade, leading to cell cycle arrest and eventual apoptosis. DNA-damaging agents, among which calicheamicins and duocarmycin derivatives are considered, act by causing strand breaks or incorporating cross-links that activate cellular death pathways. The new payload classes of pyrrolobenzodiazepines (PBDs) provide mechanisms that differ from the two above and, thus, can potentially be less amenable to cellular repair mechanisms [13].
A vital secondary mechanism by ADCs means targeting nearby cancerous cells through the bystander effect. Once released, some payloads can diffuse into adjacent cells; this could kill cancer cells which might not be expressing the target antigen. This mechanism proves extremely useful for treating heterogeneous tumours where not all cancer cells may uniformly express the target antigen, thereby extending the reach of the ADC beyond its primary targets [14].
The combined ADC results from a delicate dance between its three key constituents. The antibody determines the targeting specificity and internalisation efficacy, whereas the linker decides when and where the payload will be released. The nature of the payload stipulates both the mechanism of cell kill and the potency of cell killing. Such an integrated system must be optimised for maximum therapeutic effect while maintaining acceptable safety [15].
Table 1 highlights the multi-step process that enables ADCs to specifically target and kill cancer cells while minimizing damage to healthy tissues.
Table 2 captures some of the core advancements and strategies in the development of Antibody-Drug Conjugates (ADCs), highlighting the selection and engineering of the antibody, linker, and payload, as well as newer technologies like site-specific conjugation
| Mechanism | Description | Key Components |
|---|---|---|
| Target Recognition and Binding | ADCs use the antibody component to specifically bind to cancer cell antigens, minimizing binding to healthy tissues. Common targets include HER2 (breast cancer), CD30 (lymphomas), and CD33 (leukaemias). | Antibody, Cancer Cell Antigens |
| Cellular Internalization | The ADC is internalized by the target cell through receptor-mediated endocytosis, forming an endosome which fuses with lysosomes for payload release. | Antibody, Antigen, Endosome, Lysosome |
| Payload Release Mechanisms | Two main strategies: cleavable linkers that degrade in intracellular conditions (e.g., acidic pH, enzymes) and non-cleavable linkers that require complete antibody degradation. | Linker (Cleavable or Non-Cleavable) |
| Cytotoxic Action | Payloads include microtubule inhibitors (e.g., auristatins, maytansinoids) and DNA-damaging agents (e.g., calicheamicins, duocarmycin). Pyrrolobenzodiazepines (PBDs) offer a distinct mechanism. | Microtubule Inhibitors, DNA-Damaging Agents, PBDs |
| Bystander Effect | Some payloads can diffuse to neighboring cells, targeting and killing cancer cells that do not express the target antigen, useful in heterogeneous tumors. | Payload, Neighboring Cancer Cells |
| Integrated Mechanism | The ADC combines the antibody’s targeting specificity, the linker’s release control, and the payload’s cell-killing mechanism to optimize therapeutic effects and safety. | Antibody, Linker, Payload |
| Component | Description | Examples |
|---|---|---|
| Antibody Selection | The process involves selecting monoclonal antibodies that are humanized for compatibility and affinity-matured to enhance binding strength. Additionally, the Fc region of the antibody is altered to improve immune functions such as Antibody-Dependent Cellular Cytotoxicity (ADCC) and Complement-Dependent Cytotoxicity (CDC). | Monoclonal Antibodies, Humanized Antibodies |
| Linker Technologies | Linkers connect the antibody to the drug payload. Cleavable linkers degrade under certain intracellular conditions (e.g., acidic pH, enzymatic activities) to release the payload, while non-cleavable linkers offer stability in circulation. | Hydrazone, Peptide, Disulfide, Thioether Linkers |
| Payload Selection | The selection of potent cytotoxic agents or DNA-damaging agents is crucial. Payloads are chosen based on their ability to kill cancer cells while minimizing damage to healthy tissues efficiently. | Auristatins, Maytansinoids, DNA-Damaging Agents |
| Site-Specific Conjugation | Involves advanced methods to control where and how the antibody-drug conjugate is formed. These techniques provide better control over the drug-to-antibody ratio (DAR) and improve the uniformity and stability of the conjugate. | Cysteine Residues, Enzymatic Approaches |
| Payload Diversity | The scope of payloads has expanded beyond traditional cytotoxic agents to include immune modulators, radioisotopes, and oligonucleotides that offer new therapeutic potentials for a wider range of diseases. | Immunomodulators, Radioisotopes, Oligonucleotides |
ADCs Beyond Cancer is entering new therapeutic fields outside of oncology. In autoimmunity, ADCs are studied for the selective targeting and elimination of autoreactive immune cells in hopes of better-targeted immunosuppression in diseases like lupus and rheumatoid arthritis [16]. In infections, ADCs may help eradicate intracellular pathogens and antibiotic-resistant bacteria by delivering payloads more effectively. Research is ongoing in HIV, tuberculosis, and resistant bacterial infections [17].
The research areas include neurodegenerative diseases for delivering neuroprotective agents across the blood-brain barrier, cardiovascular diseases involving specific vascular markers in atherosclerosis, and metabolic disorders with the selective delivery of therapeutic agents [18].
To dramatically improve stability profiles and optimise pharmacokinetics. The consistency and potency of ADC products have been elevated through these methods to represent new standards in the development of targeted therapy, thus leading to more predictable results and a decrease in batch-to-batch variability [19],[20].
The development of linker technology represents one of the critical advances in ADC toward overcoming long-standing challenges associated with premature payload release and insufficient drug delivery. Such modern designs of linkers may include intricate branched structures that provide increased drug loading while ensuring stability, self-immolation mechanisms that ensure effective payload release within a desired target cell, and engineered peptide linkers that ensure circulation stability and defined intracellular cleavage patterns.
These innovations have significantly improved the therapeutic window of ADCs by reducing off-target effects while maximising drug delivery to cancer cells, leading to better clinical outcomes and fewer side effects [21].
The scope of payloads for ADCs has expanded dramatically beyond traditional cytotoxic agents with this new era of therapeutic possibilities. Currently, payload options encompass complex immune modulators that elicit targeted anti- cancer responses, precisely delivered radioisotopes that combine ADC targeting with radiotherapy benefits, and innovative therapeutic oligonucleotides capable of selective gene silencing. Such diversification has made it possible to widely expand the therapeutic potential of ADCs so that they can now be used to treat various diseases outside oncology, such as autoimmune and infectious diseases. The unprecedented versatility of the new payload classes has opened avenues that make it possible to develop particular and effective treatments across multiple therapeutic areas [10].
Recent breakthroughs in antibody engineering have introduced next-generation ADCs with bispecific and multispecific antibodies that can engage multiple targets simultaneously. These various applications highlight the ADC as a versatile platform for targeted therapy in addressing most conceivable medical challenges.
Advanced methods of site-specific conjugation have revolutionised modern ADC manufacturing and progressed far beyond the limitations of traditional random conjugation methods. From engineered cysteine residues and enzymatic approaches involving both transglutaminase and sortase A to the bright future of unnatural amino acids, a new paradigm has been established that not only works but also can provide tight control over the drug-to-antibody ratio (DAR), leading dvanced techniques in protein engineering have developed antibodies that penetrate tissues more efficiently, improve their binding characteristics, and have lower immunogenicity profiles. Engineered antibodies show high internalisation rates and maximised intracellular trafficking, significantly increasing therapeutic payloads' delivery efficiency into target cells. Moreover, advancements in new formats of antibodies have improved solid tumours' ability to penetrate better while the drug's distribution is more uniform [5]-[6].
Incorporating artificial intelligence and machine learning into ADC development has revolutionised manufacturing by allowing for the in-line optimisation of conjugation conditions and enhancing the prediction of stability profiles. Improvements in analytical techniques, including sophisticated mass spectrometry methods and continuous monitoring systems, have also significantly advanced the characterization and quality control of ADCs. These technological innovations have enhanced the production process efficiency and maintained product quality, which is of critical relevance for these complex therapeutic in the high- demand area, maintaining the highest quality when manufactured [10].
Novel approaches by formulation science have been developed to improve ADC stability and delivery. Novel excipients synthesised for ADC formulations and advanced lyophilisation techniques have improved storage stability and shelf life. Advanced drug delivery systems employing advanced technologies have allowed better control of the kinetics of the release of payloads while preserving the structural integrity of the ADC. Formulation advances in this area have dramatically contributed to the practicalities of ADC development in ensuring better stability of the product in storage and transport with preserved therapeutic efficacy [21].
Many vital areas are associated with issues and opportunities for ADC development. The major challenge in this respect is linked with mechanisms of resistance that enable cancer cells to develop mechanisms of evasion from ADC-mediated killing, including target antigen downregulation, changes in the internalisation pathways, and increased efflux of a drug [22]. Toxicity management is crucial, and efforts are made to minimise off- target effects and expand the therapeutic window of ADCs [40]. Combination therapies are also under investigation to increase effectiveness and overcome resistance, combining ADCs with immune checkpoint inhibitors, chemotherapy, or other targeted therapies [21].
Personalised medicine approaches also emerge using biomarkers and advanced diagnostics to tailor patients to the best ADC treatment [22]. Such challenges are driving innovation in ADC design and clinical strategies. To overcome resistance, researchers are now studying dual- targeting ADCs and developing novel mechanisms of action for payloads. In managing the toxicities, the approach would include more tumour-specific antibodies, optimised linker chemistry to minimise the systemic release of payloads, and careful dose optimisation.
Synergistic drug pairings are identified in combination therapy through high- throughput screening and mechanistic studies. Personalised approaches in medicine delivery are advancing further with genomic profiling, liquid biopsies, and AI-driven predictive models for guiding treatment selection and monitoring ADC [14]. These are the areas where interdisciplinary approaches can solve the ADC problems. The field of advancement in antibody engineering udes bispecific antibodies and antibody fragments that improve penetration through the tumour and reduce immunogenicity. New payload classes, including immunomodulators and DNA-modifying agents, are now being explored to increase the potential therapy for ADCs. Nanotechnology and smart delivery systems for ADCs could further enhance more precise and effectiveness of oncology. Eventually, next-generation ADCs will be developed with a better understanding of cancer biology and improved knowledge of immune responses; however, the first generation promises to be very effective and a more targeted therapy for a wider range of diseases.
Antibody-drug conjugates represent a rapidly growing class of targeted therapeutics with several approved products in the market and a strong pipeline in clinical development. Due to improved antibody engineering, linker technologies, and payload design, current ADCs show a better efficacy and safety profile than earlier generations. The outlook for further ADC development and application is promising and is likely to extend beyond oncology into other areas, such as autoimmune diseases, infectious diseases, etc. Further innovation in site-specific conjugation, new linker designs, and payload classes can only further drive ADC performance. As we better understand resistance mechanisms with the growing paradigm of personalised medicine, ADCs are destined to be increasingly integral in precision medicine, at least for many diseases where more targeted and effective treatments are urgently needed.